Research Interest
The proliferating cell nuclear antigen (PCNA) has a central role in DNA replication. Moreover, it also constitutes a central DNA scaffold for the recruitment of a plethora of components involved in both tolerance and response to DNA damage mechanisms. In eukaryotes, tolerance mechanisms are mediated by ubiquitylation of this essential replication factor PCNA, a key processivity factor for polymerases during DNA synthesis. Monoubiquitylation of Lys164 of PCNA (ubPCNAK164) enhances error-prone translesion synthesis. Further Lys63- linked polyubiquitylation of mono-ubPCNAK164 triggers the error-free mechanism based on template switching. The prevailing view is that PCNAK164 is ubiquitylated in a DNA damage-dependent manner. Therefore, it is believed that not such modification (ubPCNAK164) occurs in cells during unchallenged S-phase in most organisms except in fission yeast and Xenopus.
Although the evolutionary conserved mechanism of PCNA ubiquitylation is well understood, the deubiquitylation of ubPCNA remains uncharacterized. Our group is interested in understanding the role and, therefore, the biological significance of ubPCNA deubiquitylation in yeast models. Our working hypothesis is that Ubiquitin-specific proteases, or PCNA-DUBs, revert PCNA ubiquitylation to prevent excessive translesion synthesis on replicating chromatin. This hypothesis predicts that both branches of the DNA damage tolerance pathway, TLS-DNA polymerases sampling on replicating DNA and template switching during S-phase, would be limited at forks by PCNA deubiquitylation.
Note: Our lab is an independent Research Unit of the “Molecular Mechanism of Cancer Program” of the Cancer Research Institute (CIC-IBMCC) of the University of Salamanca (a joint Research Institute with the Spanish National Research Council or CSIC). We are also part of the Department of Microbiology and Genetics at the same University.
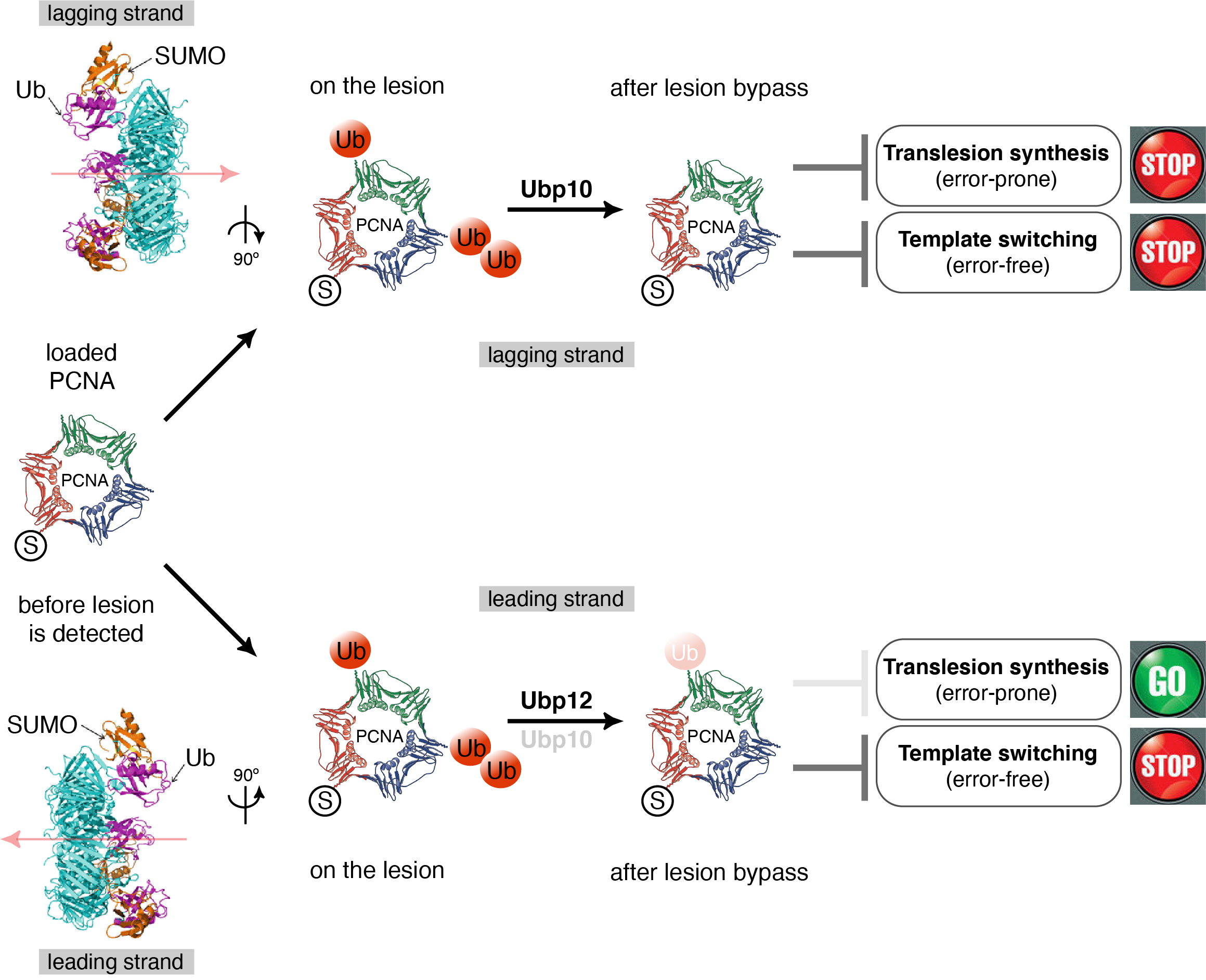
Hypothetical model for S.cerevisiae PCNA-DUBs Ubp10 and Ubp12 asymmetrical location at replication forks and potential consequences on DNA damage tolerance pathways